Global coagulation assays can be used to characterise some haemostatic disorders and to monitor the effects of treatment. Methods include viscoelastic assays, thrombin generation assays (TGA) and transmittance waveform analysis, a variation on one-stage clot assays.
Viscoelastic assays measure changes in the physiological properties of blood coagulation such as clot elasticity and rigidity, blood viscosity and platelet-mediated forces. Examples of viscoelastic assays include thromboelastic methods (i.e., thromboelastography [TEG], rotational thromboelastometry [ROTEM]), sonoclot analysis (SCT) and platelet contractile force (PCF).
Viscoelastic methods allow the assessment of whole blood samples and offer automation with an increasing level of point-of-care testing. These assays may not, however, provide a more specific profile of haemostasis than global clotting tests, with the exception of the ability to dynamically monitor coagulation.
Thromboelastic methods (i.e., TEG, ROTEM) and SCT measure all phases of coagulation from clot formation to clot retraction. PCF can measure platelet force in addition to clot structure and SCF is also useful in measuring platelet function. Viscoelastic methods generally provide a better approximation of in vivo conditions than one-stage assays and allow for the assessment of whole blood samples.
Thrombolelastic methods
The thromboelastic assays are used both to detect clot formation and to monitor changes in physiological properties, such as elasticity, over time. Key parameters measured include clotting time (CT), clot formation time (CFT) and maximal clot firmness (MCF). CT and CFT are indicative of the combined effect of coagulation factors and platelet functions, and MCF provides information about the fibrin activity in the blood sample.1,2
During TEG, a whole blood sample is placed into a temperature-regulated cup, which oscillates at a 4º angle and constant speed. A stationary pin (sensor) attached to a torsion wire is then inserted into the sample, and tissue factor (TF) and calcium are added to initiate coagulation. The fibrin that accumulates at the onset of clot development forms a mechanical connection between the pin and the cup, producing a resistance to pin rotation. The degree of resistance is then detected by a mechanical-electrical transducer and the data are transferred and analysed with specialised computer software. ROTEM is a variant of TEG in which a sensor pin attached to a shaft rotates using a ball bearing system and it is the sensor shaft, rather than the cup, that rotates.
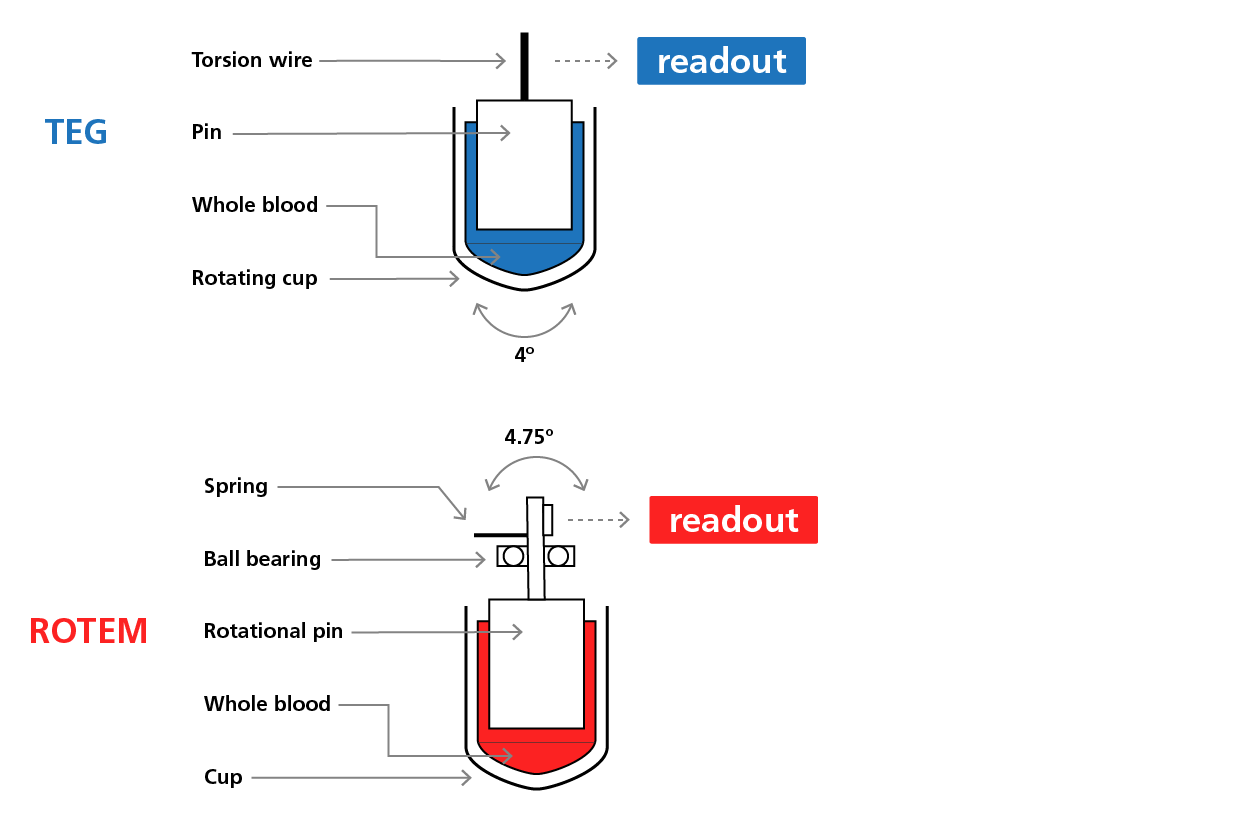
The principles behind thrombolelastography (TEG) and rotational thromboelastometry (ROTEM).1
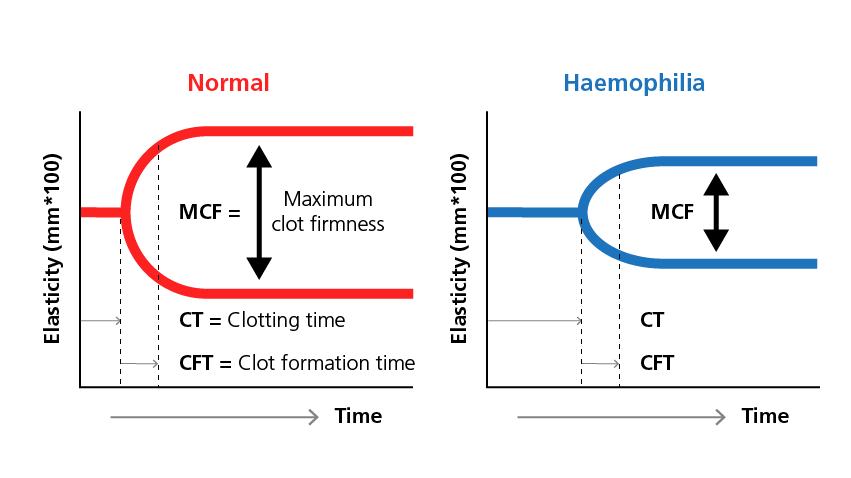
Sample thromboelastography readings in a normal patient compared to those from a patient with haemophilia.1
Sonoclot analysis (SCT)
Similar to TEG, SCT also assesses coagulation function based on changes in blood viscosity. The resulting output signal is a representation of the viscoelastic properties of clot formation. Key parameters measured include the activated clotting time (ACT), clot rate (CR), time to peak (TP), and peak amplitude (PA). ACT represents the time it takes until the onset of fibrin formation, CR is determined by the first upward slope of the sonoclot signature, the TP is determined by the second upward slope and represents the rate of conversion of fibrinogen to fibrin, and PA represents the fibrinogen concentration.3,4
During SCT, a blood sample is placed into a cuvette and a hollow probe is placed into the sample. This probe is mounted on an ultrasonic transducer and vibrates at high frequency. As the clot forms, fibrin strands assemble on the tip of the probe, producing a resistance to vibration of the probe. This increase in resistance is then detected by the probe and converted to a signal that is made visible. The resulting output signal is a representation of the viscoelastic properties of clot formation.
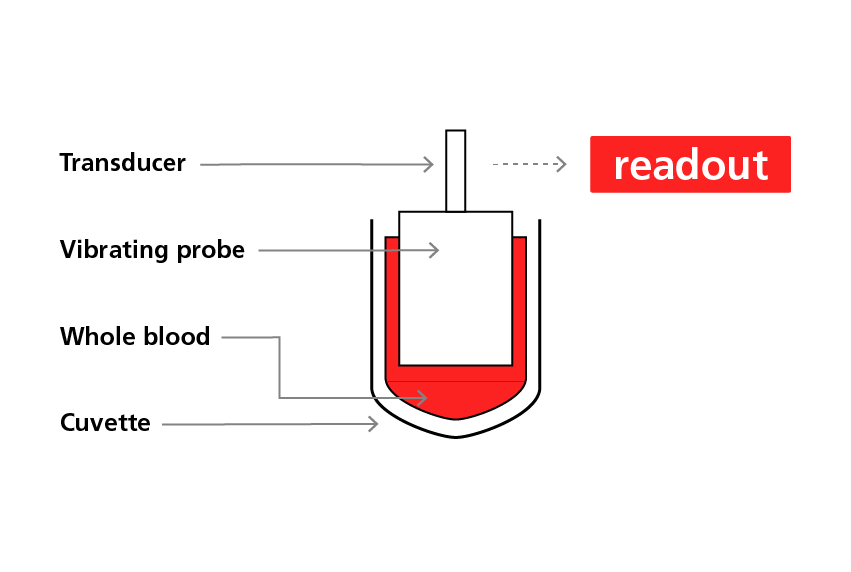
The principle behind sonoclot analysis.3,4
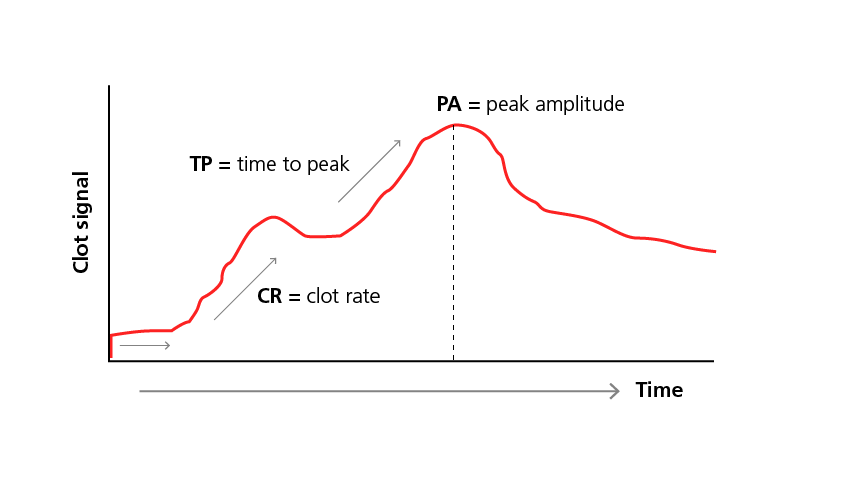
The sonoclot output signal, which is a representation of the viscoelastic properties of clot formation.3,4
Platelet contractile force (PCF)
PCF is used to determine platelet function and is based on a measurement of the platelet-mediated force developed during clotting, in contrast to SCT and thromboelastic methods, which measure clot rigidity. During the PCF test, platelets are monitored in the presence of thrombin, allowing for the assessment of platelet function during thrombin generation. This allows for the measurement of several key parameters, including PCF, the force produced by platelets during clot retraction, clot elastic modulus (CEM), and thrombin generation time (TGT). PCF is sensitive to platelet concentration and function, in addition to the presence of antithrombin activity.5
The PCF test is performed using citrated whole blood and clotting is initiated with the addition of calcium and thrombin. The sample is placed into a temperature-regulated cup and the forces produced by platelets during clot formation are measured. This is accomplished by lowering a plate into the sample and, as fibrin forms, it attaches the plate to the inner walls of the cup. Once the clot is formed, platelets within the network pull fibrin strands inward, transmitting a downward force that is transmitted to the upper plate and cup. A transducer, attached to the upper plate, measures the voltage generated in proportion to the distance moved. The PCF technique is particularly relevant in the evaluation of bleeding risks due to platelet dysfunction and in the potency assessment of anticoagulant and antiplatelet drugs.5
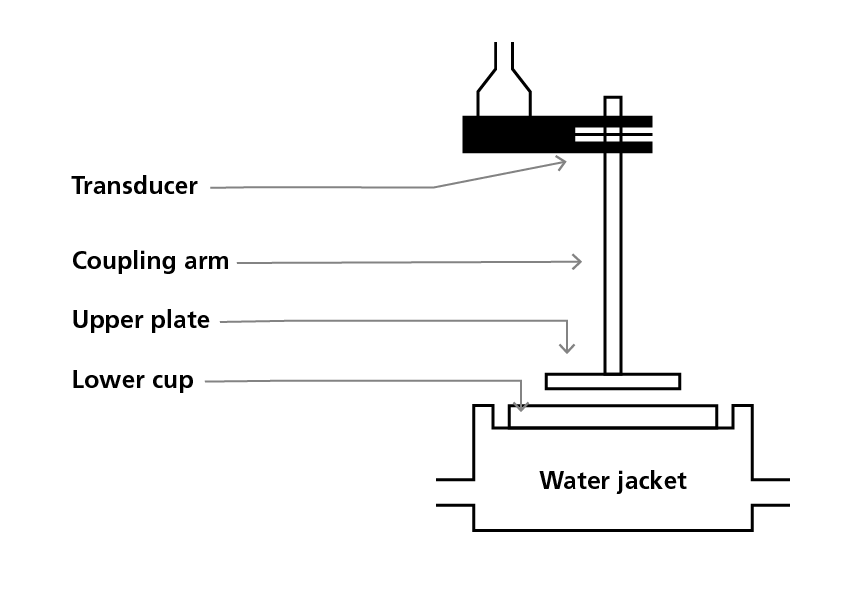
The principle behind platelet contractile force tests.5
PCF is the force produced by platelets during clot retraction. PCF is sensitive to platelet concentration and function and to the presence of antithrombin activity. If thrombin concentrations are reduced, PCF production will decrease. CEM is sensitive to fibrinogen concentration, clot structure, rate of thrombin generation and production of force by platelets. TGT is based on the kinetic profile of PCF.5
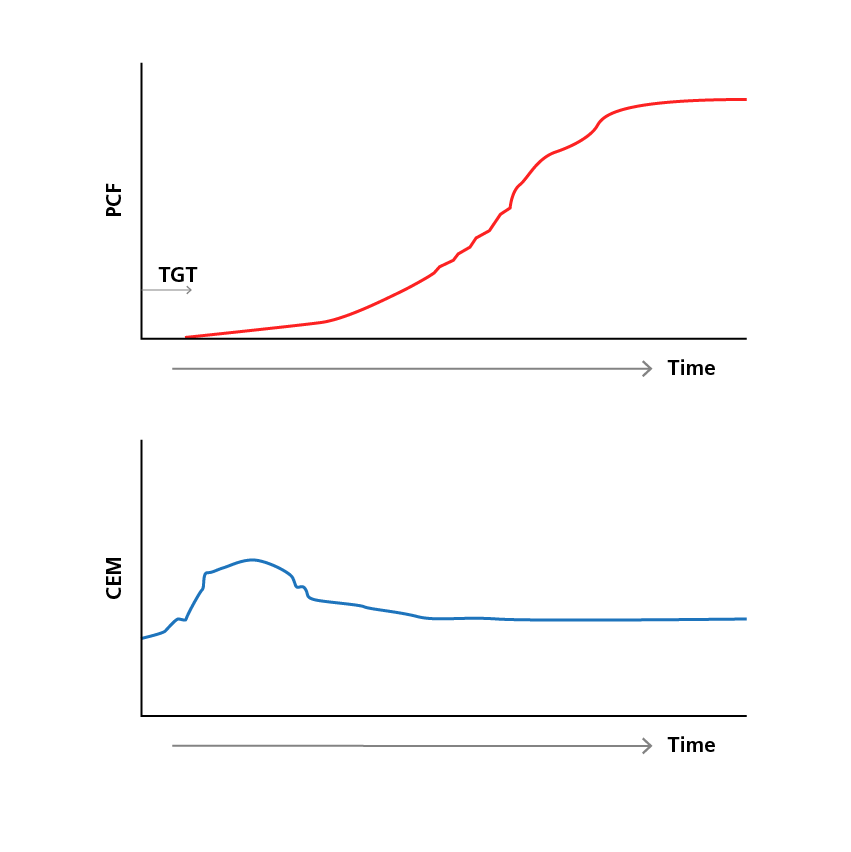
Sample platelet contractile force and clot elastic modulus readings over time.5
TGAs measure thrombin generation kinetics at all phases of the coagulation cascade, including initiation, amplification and down-regulation of thrombin generation. The assay uses a fluorogenic substrate for direct measurement of thrombin generation kinetics in platelet-rich (PRP) or platelet-poor (PPP) plasma, thereby allowing for dynamic measurements of thrombin substrate cleavage. The main parameters that can be calculated with this test include the lag time to exponential thrombin formation (initiation phase), time to peak and peak height of thrombin generation (amplification phase), and the endogenous thrombin potential (ETP).6,7
TGAs require few reagents, however the assay must be performed by a trained laboratory technician. In addition, the test is sensitive and flexible such that it can be adapted for a variety of purposes. One drawback is the need for standardised experimental conditions (currently largely missing), including TF, phospholipids and the condition of the plasma sample (i.e., frozen or fresh). This is important because lag-time, maximum thrombin concentration, and the time to reach maximum thrombin concentration are strongly influenced by the TF concentration.8
In TGAs, patient blood and reagents (including TF and phospholipids) are mixed, then calcium and a fluorogenic thrombin substrate are added as the starting reaction. The sample is analysed using a fluorogenic microtitre plate reader or with the use of an automated coagulation analyser equipped with a fluorescence measurement module. This system works by continuously monitoring the fluorescence produced by the cleavage of the fluorogenic substrate by thrombin over time. The concentration of thrombin in the sample can then be assessed by measuring the rate of changes in fluorescence intensity based on an external thrombin calibration curve or an internal calibrator.7
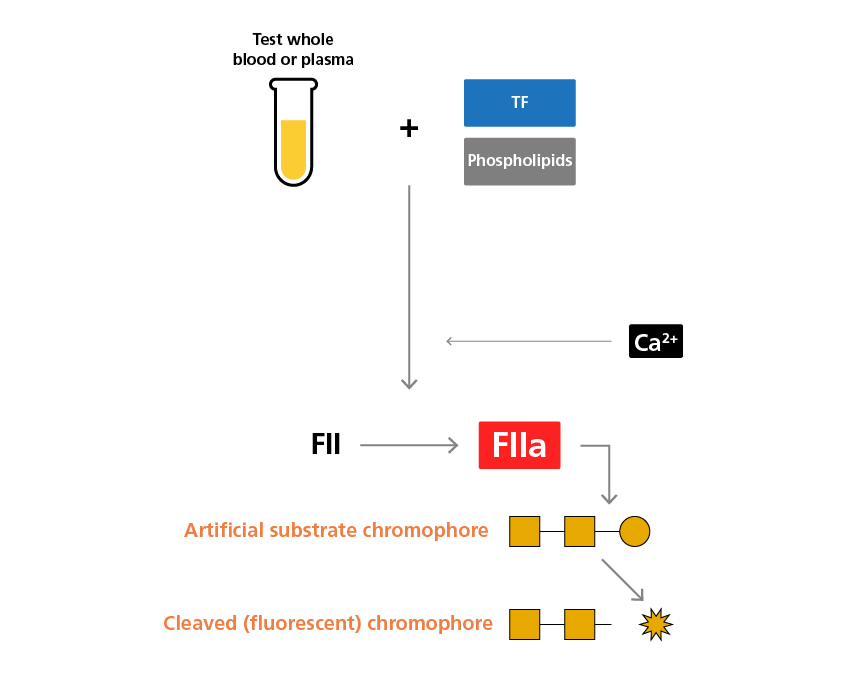
The principle behind thrombin generation assays. 7
By calculating and plotting the first derivative of the fluorescence raw signal over time, the pattern of thrombin generation in the sample for the initial phases of clot formation can be visualised. The ETP refers to the area under the curve (AUC). The ETP represents the total thrombin formed after initiation of coagulation, or the coagulation capacity.2
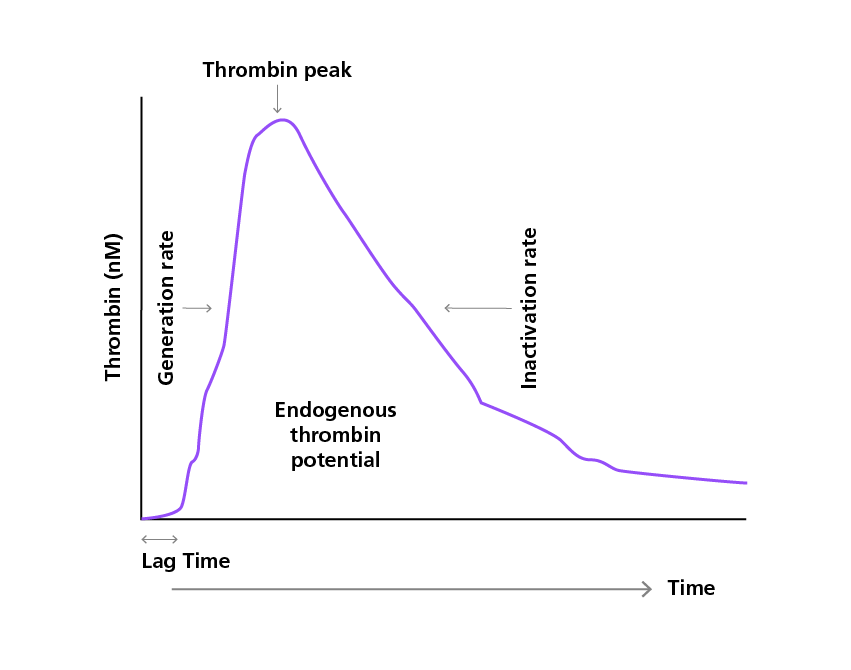
A sample thrombin generation curve in which the endogenous thrombin potential represents the total thrombin formed after initiation of coagulation.2
Calibrated automated thrombogram (CAT)
The CAT, a further development of TGA, circumvents some of the drawbacks associated with the intrinsic fluorescence of the single plasma samples by introducing a thrombin calibrator molecule (α2-macroglobulin-thrombin complexes [α2M-FIIa]). Following plasma preparation, the sample is added to different wells of the microtiter plate and either TF-reagent or the thrombin calibrator are added. The rate of thrombin activity is then calculated by comparing the fluorescent signal from the thrombin-generating sample to that from the calibrator sample with a known stable concentration of thrombin activity, measured simultaneously and in parallel, allowing for the calculation of thrombin concentration at each point of the coagulation process.9
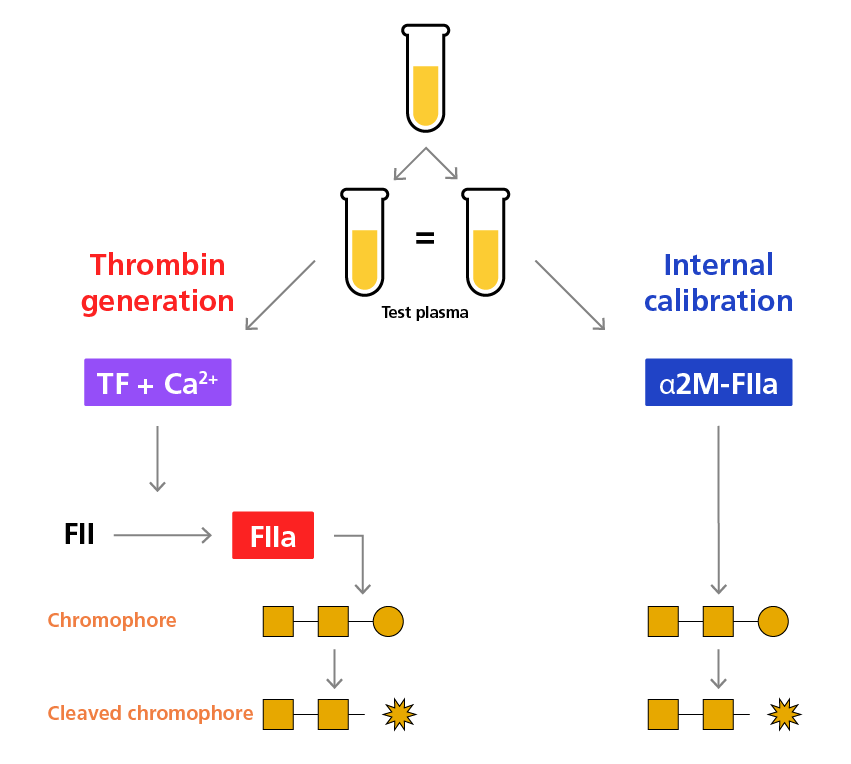
The principle behind the calibrated automated thrombogram. 9
Transmittance waveform analysis provides additional qualitative and quantitative information beyond that obtained from clotting time analysis alone. Using light transmittance, the method detects optical density changes that occur as a result of clot formation. Analysis and characterisation of clot waveform data are achieved via a photooptical coagulation analyser and can be described using a set of mathematical parameters, including clot time and the coagulation velocity. The degree of waveform anomaly usually correlates with the severity of haemostatic dysfunction.9,10
The use of activated partial thromboplastin time (aPTT) waveform analysis is particularly useful in the recognition of disseminated intravascular coagulation (DIC), even at early, non-overt stages. This assay can be performed in parallel to standard one-stage clotting tests and can be repeated as therapy progresses. Since the majority of the thrombin is generated during the later stages of the coagulation cascade and, like clotting assays, waveform analysis measures only the initial stage of clot formation, the assay is not able to assess the contribution of platelets and phospholipids, nor assess the status of hyper- and hypocoagulation.10,11
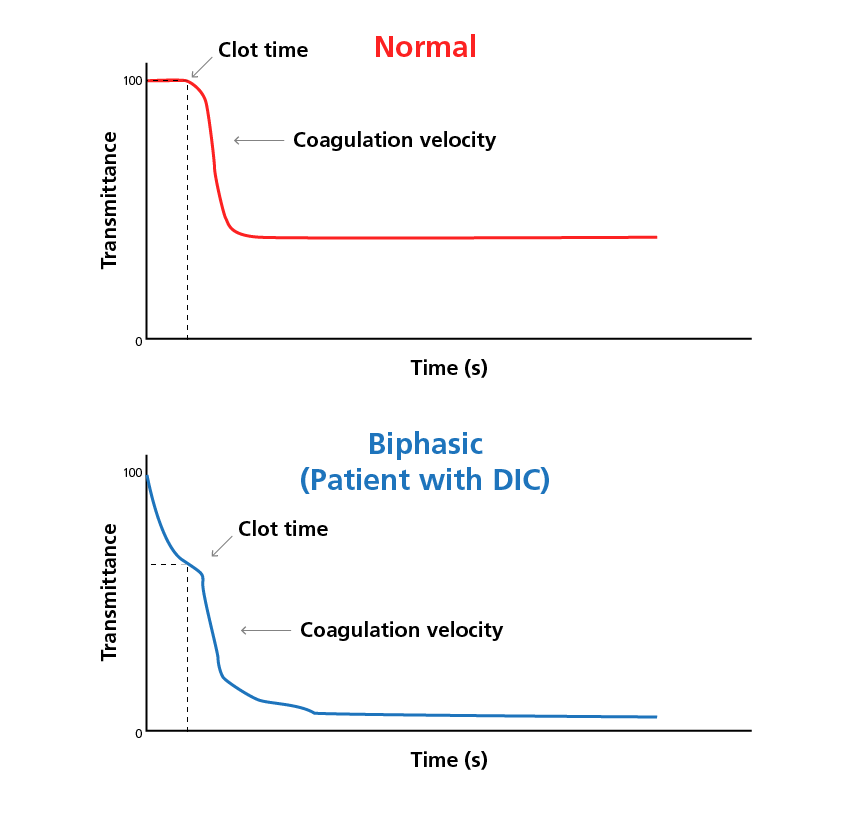
Sample activated partial thromboplastin time clot waveform readings in a normal patient and from a patient with disseminated intravascular coagulation. 10
1. Gabriel DA, Carr M, Roberts HR. Monitoring coagulation and the clinical effects of recombinant factor VIIa. Semin Hematol 2004;41:20-4.
2. Varadi K, Turecek PL, Schwarz HP. Thrombin generation assay and other universal tests for monitoring haemophilia therapy. Haemophilia 2004;10 17-21.
3. Furuhashi M, Ura N, Hasegawa K, et al. Sonoclot coagulation analysis: new bedside monitoring for determination of the appropriate heparin dose during haemodialysis. Nephrol Dial Transplant 2002;17:1457-62.
4. Hett DA, Walker D, Pilkington SN, Smith DC. Sonoclot analysis. Br J Anaesth 1995;75:771-6.
5. Carr ME, Jr. Development of platelet contractile force as a research and clinical measure of platelet function. Cell Biochem Biophys 2003;38:55-78.
6. Chantarangkul V, Clerici M, Bressi C, Giesen PL, Tripodi A. Thrombin generation assessed as endogenous thrombin potential in patients with hyper- or hypo-coagulability. Haematologica 2003;88:547-54.
7. Hemker HC, Willems GM, Beguin S. A computer assisted method to obtain the prothrombin activation velocity in whole plasma independent of thrombin decay processes. Thromb Haemost 1986;56:9-17.
8. Gerotziafas GT, Depasse F, Busson J, Leflem L, Elalamy I, Samama MM. Towards a standardization of thrombin generation assessment: the influence of tissue factor, platelets and phospholipids concentration on the normal values of Thrombogram-Thrombinoscope assay. Thromb J 2005;3:16.
9. Hemker HC, Giesen P, AlDieri R, et al. The calibrated automated thrombogram (CAT): a universal routine test for hyper- and hypocoagulability. Pathophysiol Haemost Thromb 2002;32:249-53.
10. Toh CH, Giles AR. Waveform analysis of clotting test optical profiles in the diagnosis and management of disseminated intravascular coagulation (DIC). Clin Lab Haematol 2002;24:321-7.
11. Toh CH. Transmittance waveform of routine coagulation tests is a sensitive and specific method for diagnosing non-overt disseminated intravascular coagulation. Blood Rev 2002;16 Suppl 1:S11-4.