Antigen-based assays can be used to distinguish quantitative (reduced protein expression or secretion) from qualitative (functional) deficiencies. Two main types of antigen-based assays are commonly used: the enzyme-linked immunosorbent assay (ELISA) and the latex immunoassay. The ELISA uses a specific antibody to capture an antigen and a second enzyme-linked antibody to measure the amount of antigen, whereas latex immunoassays are single- antibody systems.
The von Willebrand factor/factor VIII-binding (VWF-FVIII-binding) assay is used to distinguish the 2N subtype from other VWD type 2 subtypes, and von Willebrand factor multimer analysis is typically performed after initial VWD assays to help define the VWD variant subtype. Clotting factor antigen assays may be used to differentiate functional from quantitative defects.
Sandwich enzyme-linked immunosorbent assays (ELISA) are a common antigen-based assay format, in which a polyclonal antibody that recognises the coagulation factor to be assessed is coated on a solid surface, typically microplate wells. Plasma samples are diluted and applied to the surface, where the antigen, if present, is recognised by the polyclonal antibody. Following washing steps to remove unbound antigen, the same polyclonal antibody, with a colourimetric label attached, is permitted to bind to the substrate. The complex is washed again and the colourimetric label is detected and quantified using a spectrophotometer. Diluted control plasma comprising a known concentration of wild-type antigen serves as the comparator and calibrator.
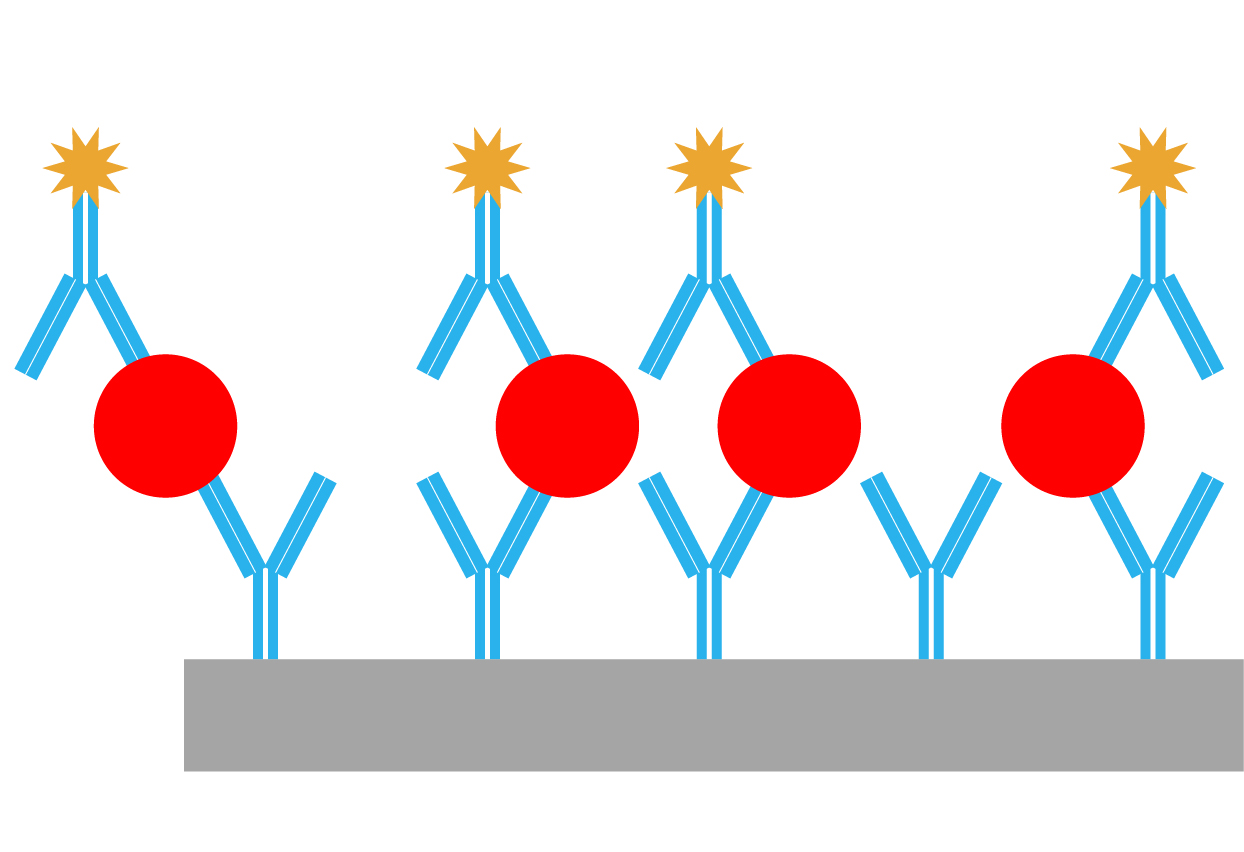
The principle behind enzyme-linked immunosorbent assays (ELISA)
Latex particles coated with antibody specific to an antigen agglutinate in the presence of that antigen. The degree of agglutination is directly proportional to the concentration of the antigen in the sample and is determined by measurement of transmitted light or light scattering.
Factor (F) XIII antigen assays are recommended by the Factor XIII and Fibrinogen SSC Subcommittee of the International Society on Thrombosis and Haemostasis (ISTH) as a second step to determine the subtype of FXIII deficiency (FXIII-A or FXIII-B deficiency) subsequent to having detected a decrease in FXIII activity.5 If plasma FXIII-A2B2 concentration is decreased, detection of FXIII-A and FXIII-B antigens should follow.5-7 FXIII antigen concentrations can be measured using a sandwich enzyme-linked immunosorbent assay (ELISA) or latex immunoassay.6,8,9
To detect plasma FXIII-A2B2, for example, a
biotinylated monoclonal capture antibody against FXIII-B that is
immobilised in streptavidin-coated wells and a horseradish peroxidase
(HRP)-labelled
monoclonal detection antibody against FXIII-A are used.10
To detect FXIII-A or FXIII-B subunits, both the biotinylated capture
and the HRP-labelled detection antibodies react with only one of the
two subunits (FXIII-A or FXIII-B), but are directed against different
epitopes of the same antigen molecule.10
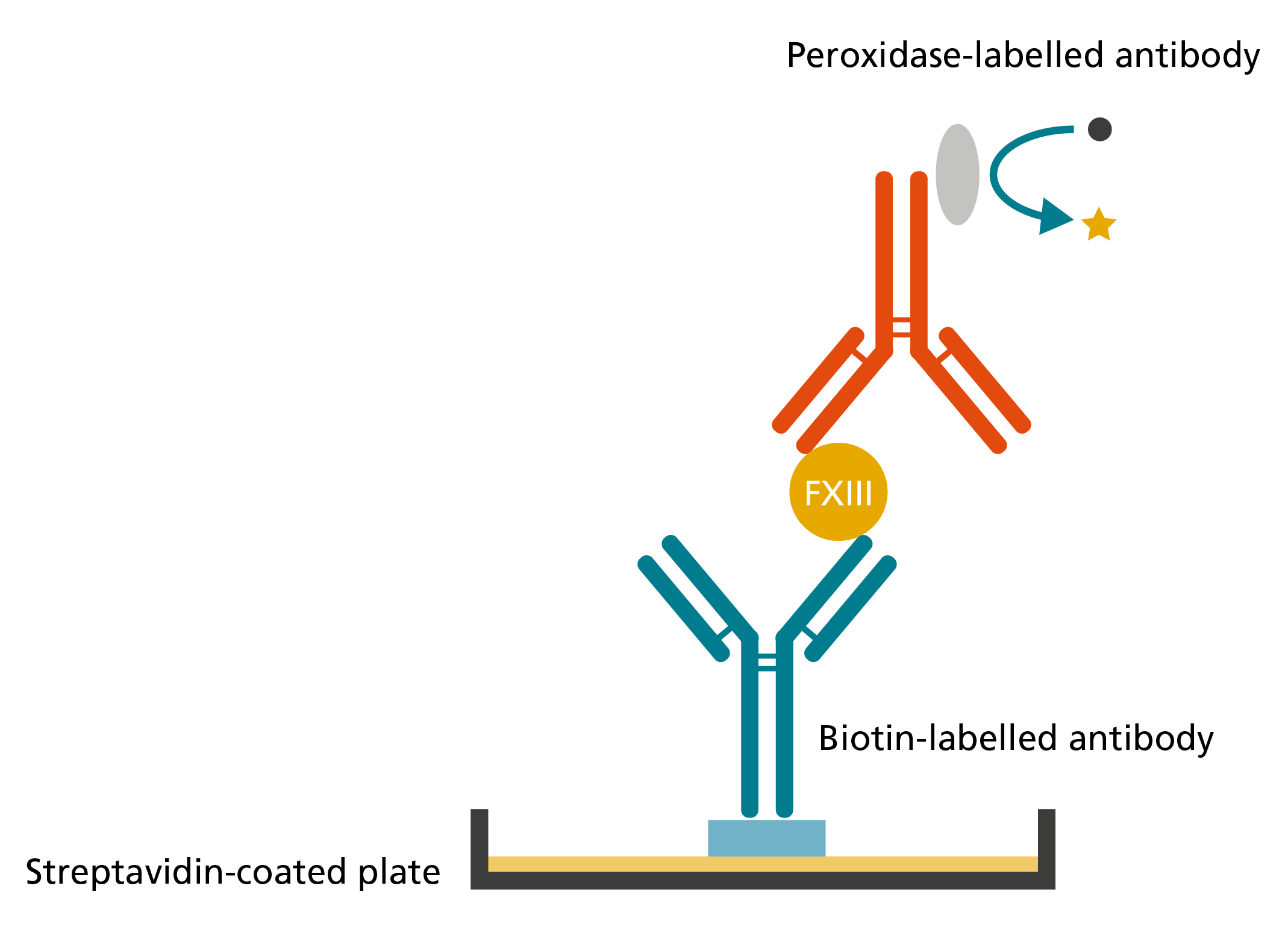
Example that demonstrates the principle of an ELISA system detecting FXIII antigen.11
Latex immunoassay
Latex particles coated with antibody specific to human FXIII agglutinate in the presence of FXIII. The degree of agglutination is directly proportional to the concentration of FXIII antigen in the sample and is determined by measurement of transmitted light or light scattering (see product information of respective assays). Commercially available latex immunoassays include HemosIL® Factor XIII Antigen (Werfen, Instrumentation Laboratory, Bedford, MA, USA) specific for FXIIIA and K-Assay Factor XIII (Stago, Asnières sur Seine, France; Kamiya Biomedical Company, Seattle, WA, USA).
1. Nichols WL, Hultin MB, James AH, et al. von Willebrand disease (VWD): evidence-based diagnosis and management guidelines, the National Heart, Lung, and Blood Institute (NHLBI) Expert Panel report (USA). Haemophilia 2008;14:171-232.
2. James AH, Eikenboom J, Federici AB. State of the art: von Willebrand disease. Haemophilia 2016;22 Suppl 5:54-9.
3. Asselta R, Peyvandi F. Factor V deficiency. Semin Thromb Hemost 2009;35:382-9.
4. Girolami A, Cosi E, Sambado L, Girolami B, Randi ML. Complex history of the discovery and characterization of congenital factor X deficiency. Semin Thromb Hemost 2015;41:359-65.
5. Kohler HP, Ichinose A, Seitz R, et al. Diagnosis and classification of factor XIII deficiencies. J Thromb Haemost 2011;9:1404-6.
6. Biswas A, Ivaskevicius V, Thomas A, Oldenburg J. Coagulation factor XIII deficiency. Diagnosis, prevalence and management of inherited and acquired forms. Hamostaseologie 2014;34:160-6.
7. Bolton-Maggs PH, Favaloro EJ, Hillarp A, Jennings I, Kohler HP. Difficulties and pitfalls in the laboratory diagnosis of bleeding disorders. Haemophilia 2012;18 Suppl 4:66-72.
8. Schroeder V, Kohler HP. Factor XIII deficiency: an update. Semin Thromb Hemost 2013;39:632-41.
9. Opat S, Butler J, Malan E, Duncan E, Tran HA. Factor XIII assays. Methods Mol Biol 2013;992:171-80.
10. Orosz ZZ, Katona E, Facsko A, Berta A, Muszbek L. A highly sensitive chemiluminescence immunoassay for the measurement of coagulation factor XIII subunits and their complex in tears. J Immunol Methods 2010;353:87-92.
11. Katona E, Haramura G, Karpati L, Fachet J, Muszbek L. A simple, quick one-step ELISA assay for the determination of complex plasma factor XIII (A2B2). Thromb Haemost 2000;83:268-73.
12. Nichols WL, Hultin MB, James AH, et al. von Willebrand disease (VWD): evidence-based diagnosis and management guidelines, the National Heart, Lung, and Blood Institute (NHLBI) Expert Panel report (USA). Haemophilia 2008;14:171-232.
13. James AH, Eikenboom J, Federici AB. State of the art: von Willebrand disease. Haemophilia 2016;22 Suppl 5:54-9.