Chromogenic activity assays use coloured or fluorescent substrates to quantify the enzymatic activity or function of specific coagulation factors. The assays function on the assumption that the coloured or fluorescent product produced is proportional to the level of factor activity in the sample.
Chromogenic FVII activity assays are based on the same principle as clot-based FVII assays, the only important difference being the endpoint used to quantify the factor activity. Two types of chromogenic assay have been developed; both use a FXa-specific substrate to quantify upstream enzymatic activity.
In the conventional version of the FVII chromogenic assay, FVII zymogen is first converted to FVIIa in the presence of thromboplastin and calcium, then the total FVII(a) concentration is measured as a function of FXa activity. A more recent version of the FVII chromogenic assay specifically measures FVIIa activity in a citrated plasma sample. A recombinant, truncated soluble tissue factor (sTF) binds to FVIIa, but is not able to activate FVII zymogen. In the presence of phospholipids and calcium, the sTF-FVIIa complex activates FX and FXa cleaves a specific peptide substrate. Another variant of this assay uses a FVIIa-specific antibody. FXa activity in the assay is directly proportional to FVIIa activity. The use of synthetic phospholipids has reduced interference from impurities and improved assay specificity, particularly in the low concentration range.1
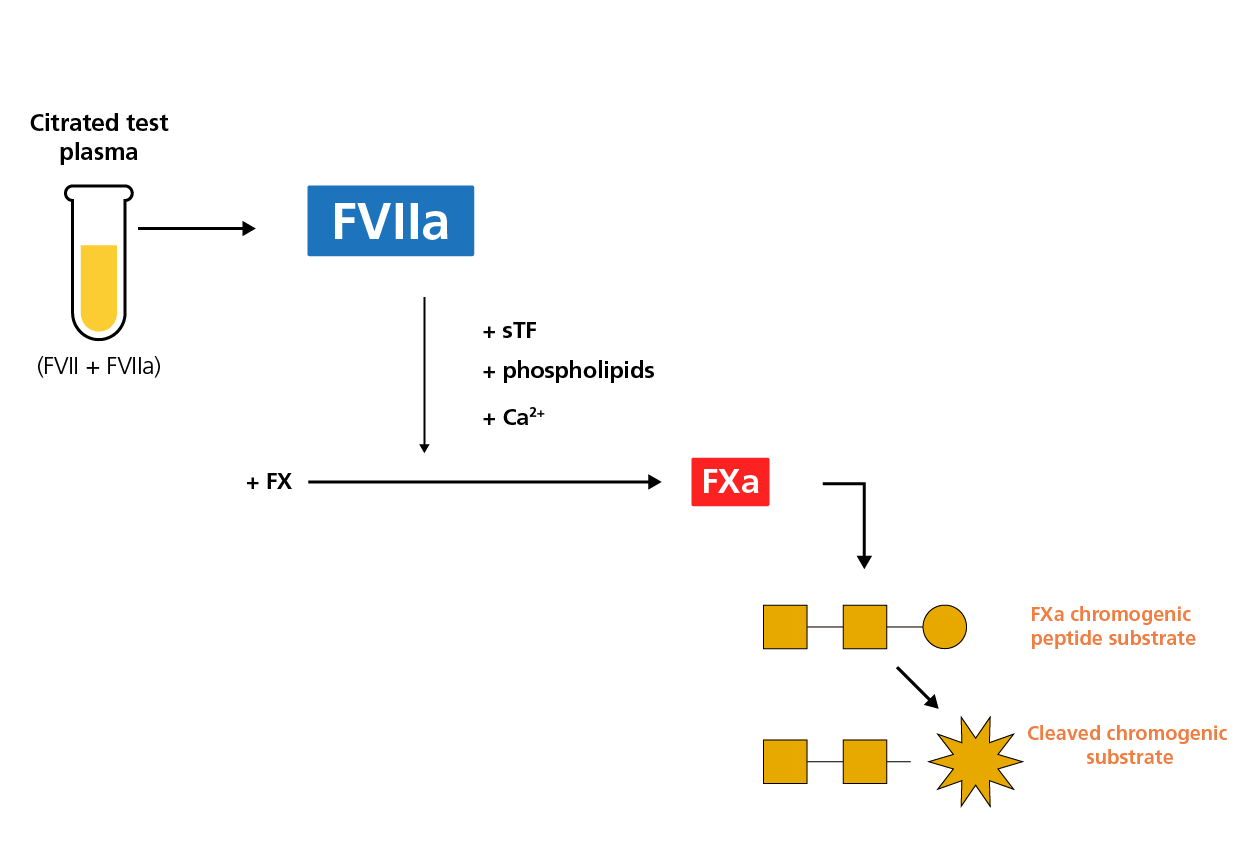
A schematic diagram of the chromogenic FVIIa assay.
Chromogenic FVIII activity assays are derived from two-stage clotting assays2,3 and can be used to measure the functional activity of FVIII. In the chromogenic FVIII activity assay, the first stage is comprised of mixing the test plasma with thrombin (FIIa) or prothrombin (FII), FIXa, FX, phospholipids and calcium, which leads to the activation of FX (FXa). During the second stage, a thrombin inhibitor and a chromogenic substrate specific for FXa are added, and FXa activity levels are quantified by monitoring the cleavage of the chromogenic peptide substrate.4
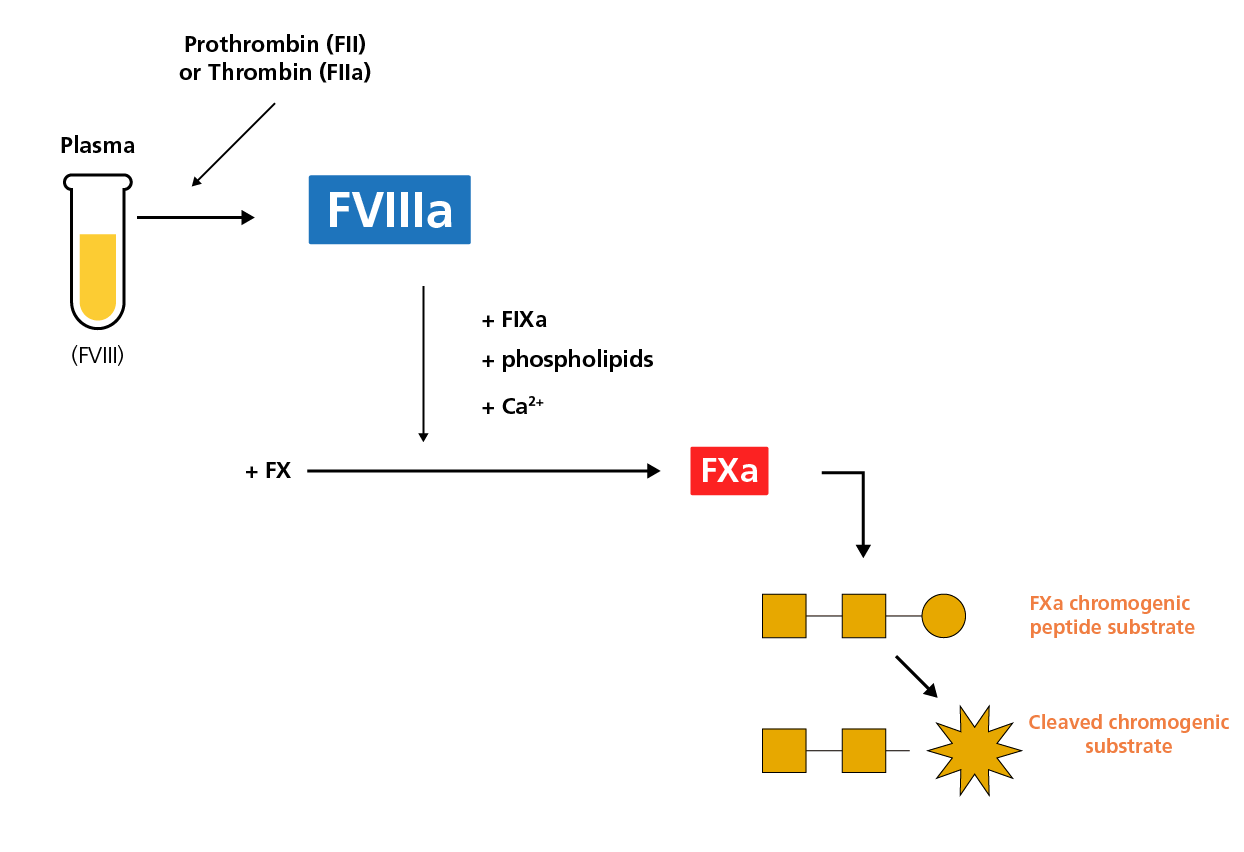
A schematic diagram of the chromogenic FVIII assay.
In the chromogenic FIX activity assay, the first stage is comprised of mixing the test plasma with FXIa, FVIII, thrombin (FIIa), prothrombin (FII), FX, phospholipids and calcium, which leads to the FIX-dependent activation of FX (FXa). During the second stage, an off-target (non-FXa) enzyme inhibitor and a chromogenic substrate specific for FXa are added, and FXa activity levels are quantified by monitoring the cleavage of the chromogenic peptide substrate.4
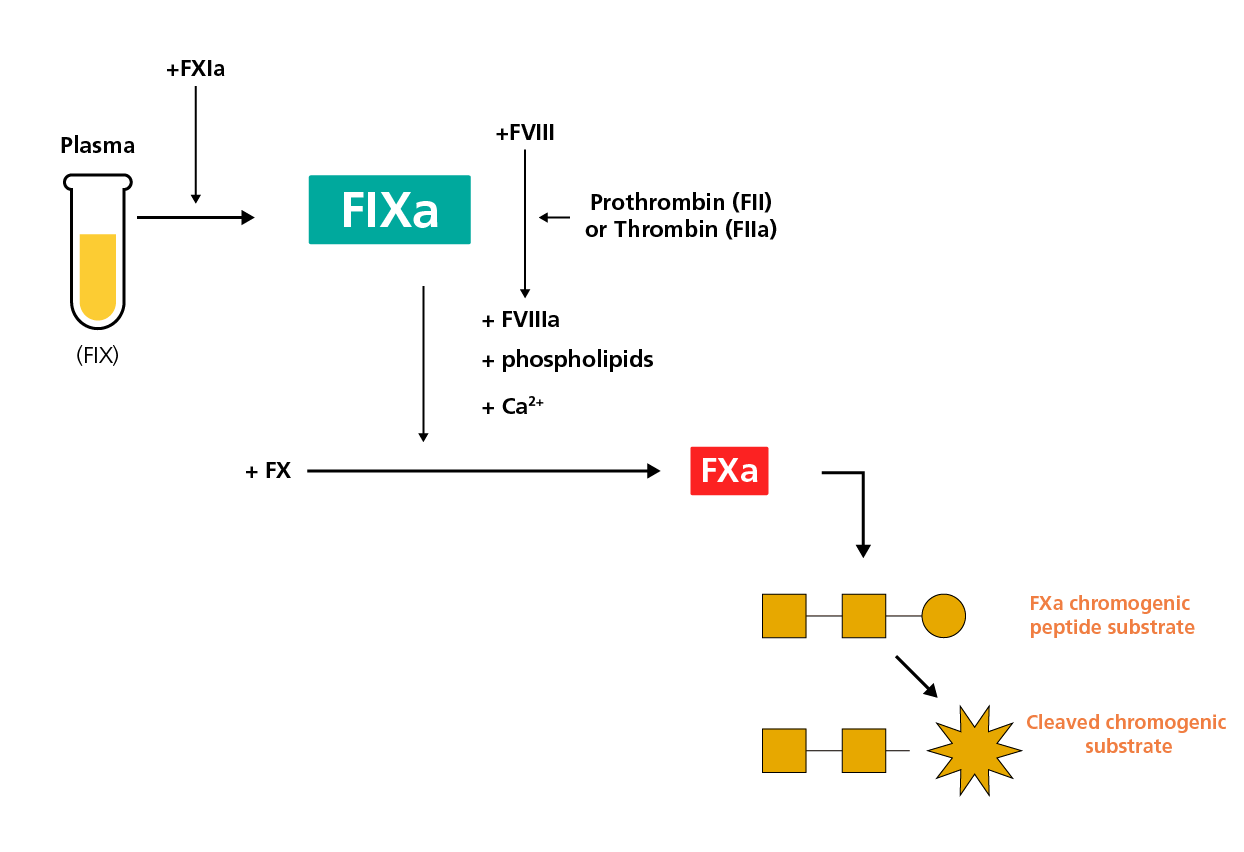
A schematic diagram of the chromogenic FIX assay.
FXIII activity assays are recommended as a “first-line” test to screen for FXIII deficiency by the Factor XIII and Fibrinogen SSC Subcommittee of the International Society on Thrombosis and Haemostasis (ISTH).5 They are based on two principles: the measurement of ammonia release during the first step of the transglutaminase reaction and the measurement of labelled amine covalently cross-linked to a protein substrate by activated FXIII (FXIIIa).5-8 The newer fluorometric assay is based on the isopeptidase activity of FXIIIa.7,8
Ammonia release assay
In the ammonia release assay, ammonia produced during the transglutaminase reaction mediated by FXIIIa is spectrophotometrically measured by a glutamate dehydrogenase (GIDH)-mediated indicator reaction in which nicotinamide adenine dinucleotide phosphate (NADPH) is converted to NADP.5,7,8 Initially, FXIII in the plasma is activated by thrombin and Ca2+ and the clotting of plasma inhibited by a fibrin polymerisation inhibitory peptide.5,8 FXIIIa then cross-links an amine substrate to a glutamine residue in an oligopeptide, whereby ammonia is released.5,8

1. Activation during the lag phase:
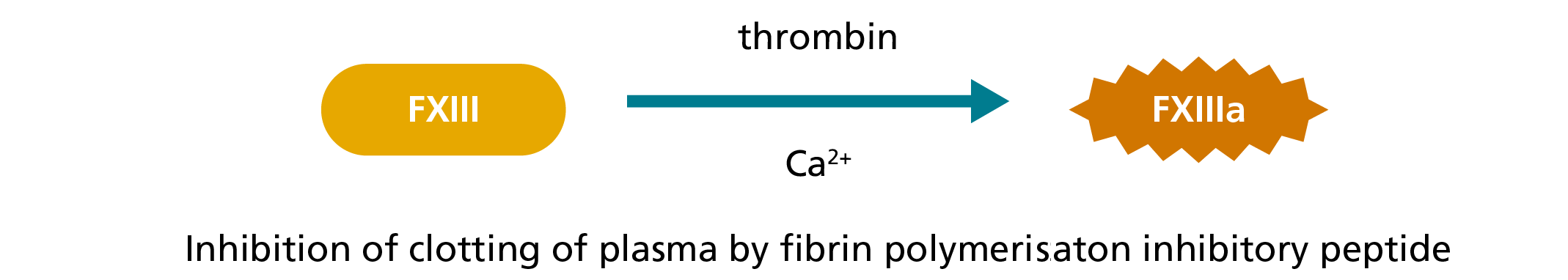
2. Transglutaminase reaction:

3. Monitoring the release of ammonia:
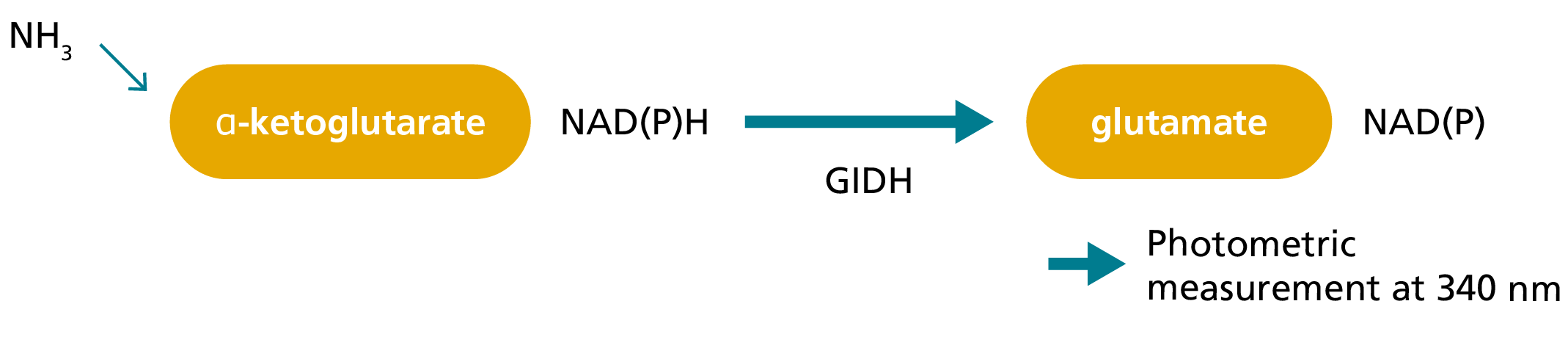
Principle of photometric ammonia release assay to measure FXIII activity.8
Because other NADPH-consuming and ammonia-producing reactions that are independent of activated FXIII are present in plasma, overestimation of FXIII activity can potentially occur, a problem that is especially significant in the low activity range.5,7,8 Therefore, a parallel plasma blank sample is required and the blank value must be subtracted from the test value in order to precisely determine FXIII activity.5,7,8 Another disadvantage of ammonia release assays is that they have a relatively low sensitivity with a limit of quantitation between 3% and 5%.5,8
Amine incorporation assay
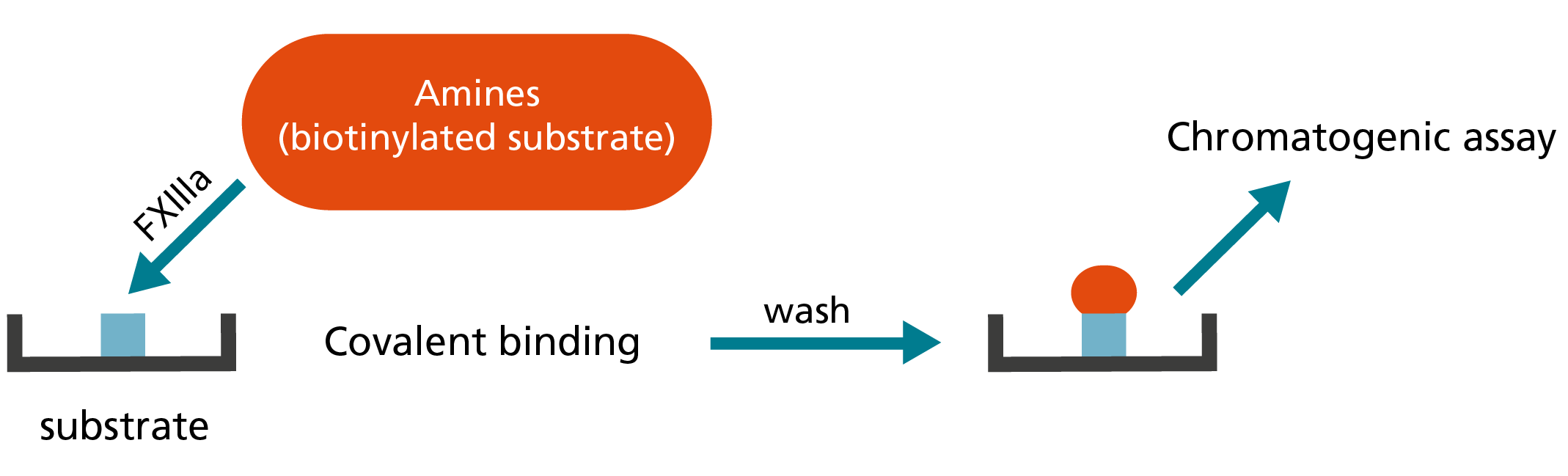
In the amine incorporation assay, fluorescent, radiolabelled or biotinylated amine substrates are covalently bound to a glutamine residue of a protein substrate by FXIIIa. Free unbound amine substrate molecules are then separated from the protein-bound fraction.5,8 The latter is quantitated by measuring the incorporated fluorescent or radiolabelled amine substrates or by using a streptavidin-mediated enzyme reaction in case of biotinylated amines.8 As in the ammonia release assay, the initial FXIII activation is mediated by thrombin and Ca2+.5
1. Activation during course of reaction:
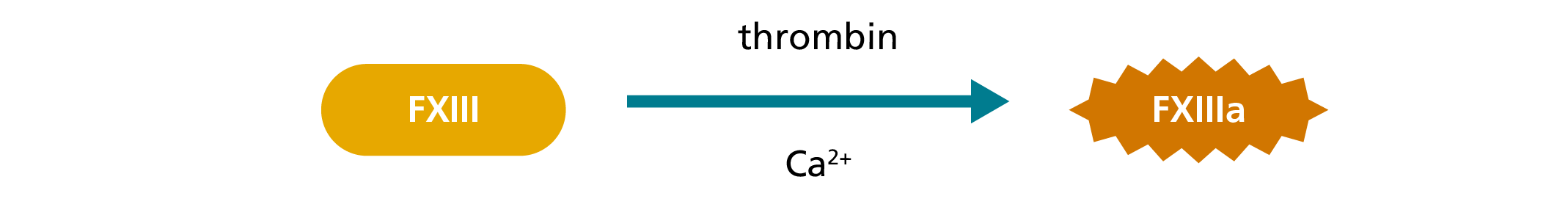
2. Transglutaminase reaction:
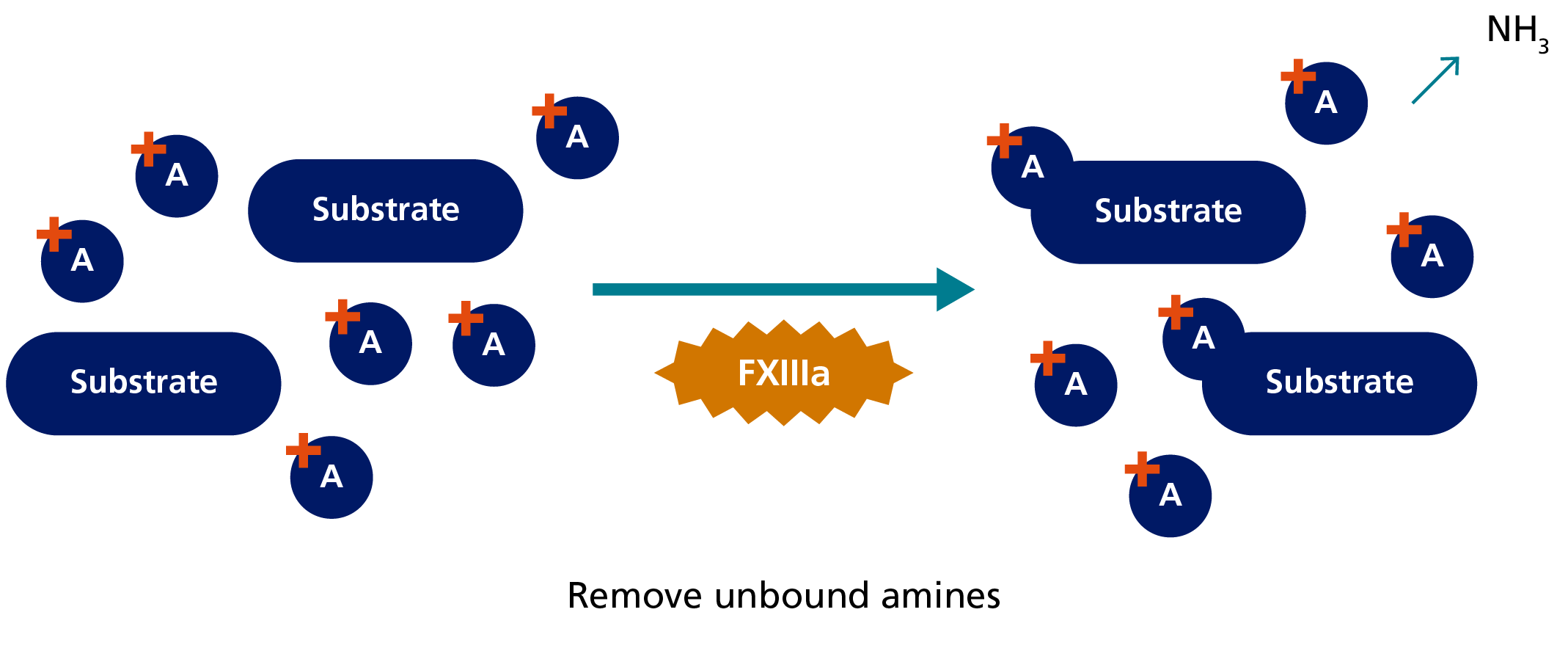
3. Determination of incorporated amine:

Principle of amine incorporation assay to measure FXIII activity.
Amine incorporation assays were the first quantitative FXIII assays used to determine FXIII activity and are very sensitive, some being able to detect plasma FXIII activity levels below 1 IU/dL (%).8 However, their disadvantage is that they are rather time-consuming, difficult to standardise and the separation step makes it hardly possible to design a true kinetic assay.5,8 Furthermore, the FXIII-A Val34Leu polymorphism common in the Caucasian population can influence those types of amine incorporation assays that measure transglutaminase activity at the initial stage of FXIII activation.5,8 In contrast, amine incorporation assays that measure the transglutaminase activity of the fully activated enzyme are not affected by this polymorphism.5,8
Fluorometric assays
The newer fluorometric assay is based on the isopeptidase activity of activated FXIII under certain conditions in which the enzyme can release primary amines bound to a glutamine residue in an oligopeptide.8 The gradual removal of the amine, which contains a quencher, leads to an increased fluorescence of the peptide substrate, which is labelled with a fluorophore.8 However, compared to ammonia release assays, the fluorometric assay has an even lower sensitivity and assay results are not well correlated.8
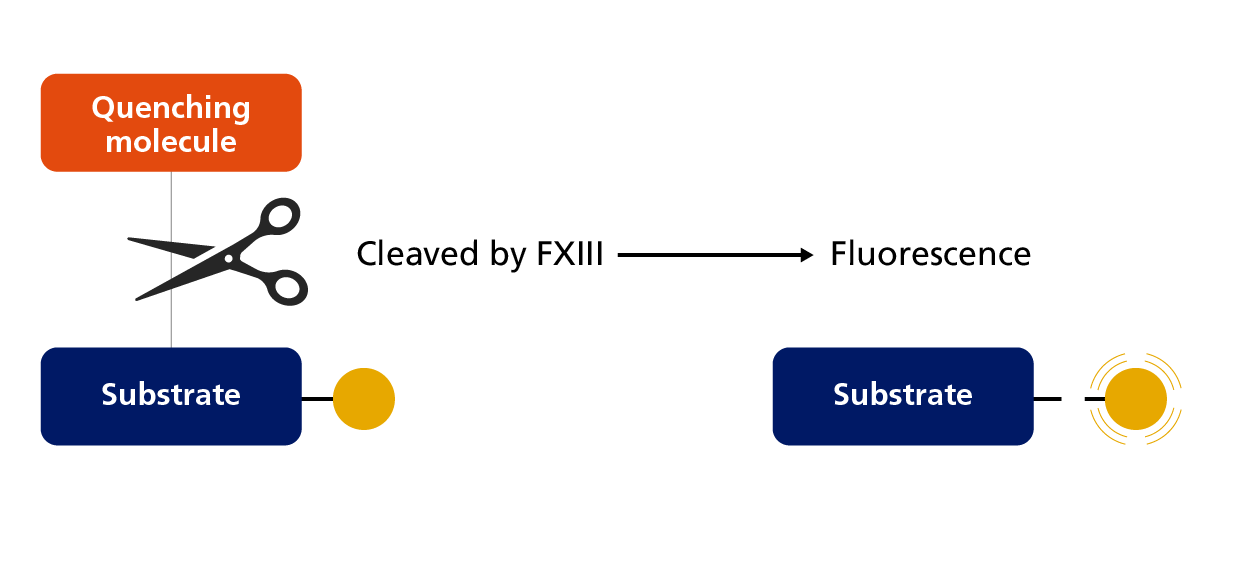
Principle of the fluorometric isopeptidase assay to measure FXIII activity.
1. Amiral J, Dunois C, Amiral C, Seghatchian J. The various assays for measuring activity states of factor VIIa in plasma and therapeutic products: Diagnostic value and analytical usefulness in various pathophysiological states. Transfus Apher Sci 2017;56:91-7.
2. Moser KA, Adcock Funk DM. Chromogenic factor VIII activity assay. Am J Hematol 2014;89:781-4.
3. Biggs R, Eveling J, Richards G. The assay of antihaemophilic-globulin activity. Br J Haematol 1955;1:20-34.
4. Bates SM, Weitz JI. Coagulation assays. Circulation 2005;112:e53-60.
5. Kohler HP, Ichinose A, Seitz R, et al. Diagnosis and classification of factor XIII deficiencies. J Thromb Haemost 2011;9:1404-6.
6. Bolton-Maggs PH, Favaloro EJ, Hillarp A, Jennings I, Kohler HP. Difficulties and pitfalls in the laboratory diagnosis of bleeding disorders. Haemophilia 2012;18 Suppl 4:66-72.
7. Schroeder V, Kohler HP. Factor XIII deficiency: an update. Semin Thromb Hemost 2013;39:632-41.
8. Katona E, Penzes K, Molnar E, Muszbek L. Measurement of factor XIII activity in plasma. Clin Chem Lab Med 2012;50:1191-202.