
Platelets are a key component of the haemostatic process, with aggregated platelets forming a platelet plug at the site of tissue injury, which then serves as a surface and source of phospholipids for other factors in the coagulation cascade. In the absence of adequate platelet aggregation and clot retraction, due to absent or dysfunctional glycoprotein IIb/IIIa, patients with Glanzmann’s thrombasthenia (GT) are at risk of bleeding.1
Patients with GT typically present with a normal platelet count and morphology, and normal prothrombin time (PT) and activated partial thromboplastin time (aPTT), but have a prolonged bleeding time, which should warrant further investigation, largely comprising platelet function tests. A personal or familial history of bleeding and easy bruising may also support the diagnosis. The gold standard for the differential diagnosis of GT is the specific aggregation of platelets in the presence of the antibiotic ristocetin, which is fibrinogen-independent, but not in the presence of other physiological agonists. Confirmation can be achieved using further platelet function assays and characterisation of the molecular and genetic lesion.1-4
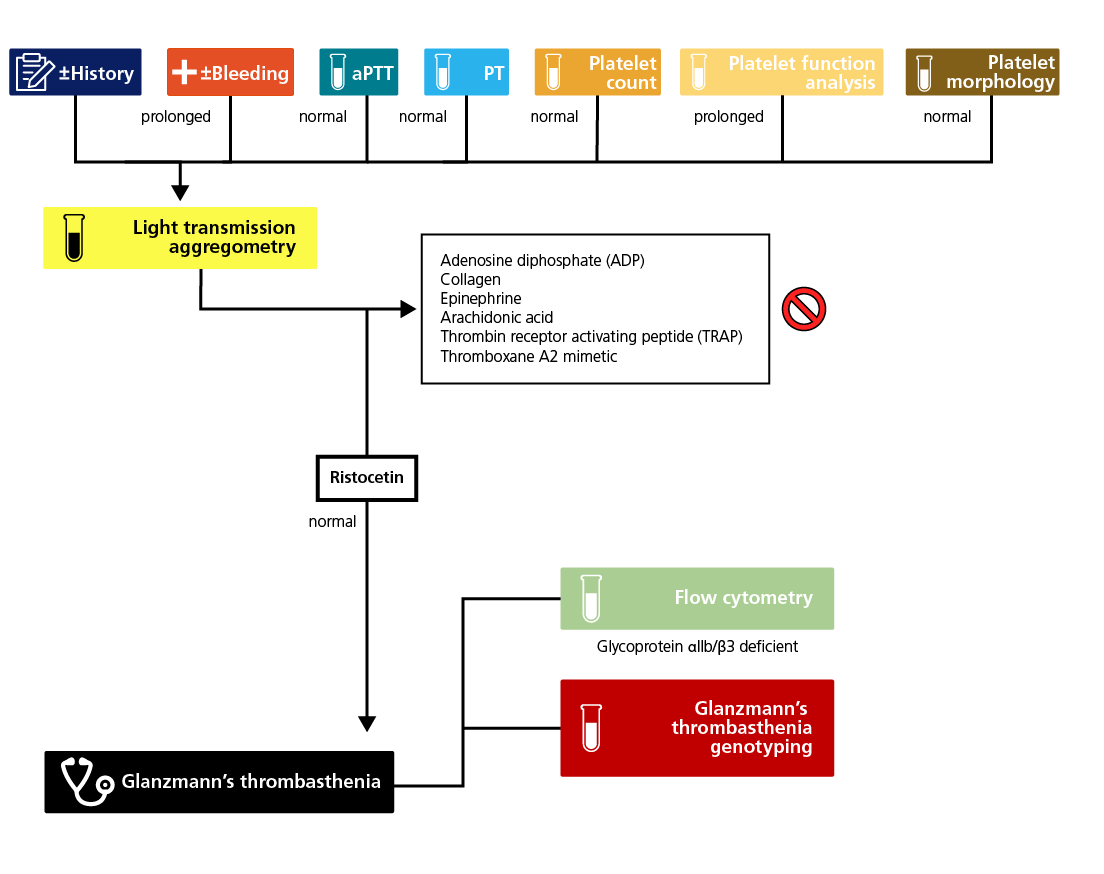
Algorithm for the laboratory diagnosis of Glanzmann’s thrombasthenia (GT).
Initial laboratory screening
In patients with GT, platelet count and morphology are likely to be normal, as are PT and aPTT, while bleeding time is prolonged.
Platelet function analysis (PFA)
PFA is also highly sensitive for GT and investigates the ability of platelets in citrated whole blood to bind to cartridges comprising collagen and ADP or epinephrine, which are intended to imitate a damaged blood vessel, at a high shear stress rate. Samples from patients with GT are unable to form a platelet plug. This assay serves as a superior alternative to bleeding time.1
Light transmission aggregometry (LTA)
LTA serves as a highly-specific assay for GT. Platelet aggregation in the presence of physiological agonists, including adenosine diphosphate (ADP), collagen, epinephrine, arachidonic acid, thrombin receptor activating peptide (TRAP), thromboxane A2 mimetic and ristocetin, is tested using platelet-rich plasma. Among these agents, ristocetin alone is able to induce aggregation in GT-affected samples because it can act in a fibrinogen-independent manner. Challenges include access to specialist laboratory capabilities and the need for platelet-rich plasma in patients with thrombocytopenia or limited permissible blood sampling volume.1
Flow cytometry
Flow cytometry uses monoclonal antibodies to assess the density of specific receptors or other antigens on the platelet surface and can provide additional information about a specific deficiency in glycoprotein (GP)IIb (CD41) and GPIIIa (CD61) expression or function. This method is useful for rapid analysis of the carrier status for potentially-affected family members or as part of a pre-natal diagnosis.1,3,4
Molecular analysis (genotyping)
A molecular genetic analysis of the ITGA2B and ITGB3 genes, encoding GPIIb and GPIIIa, respectively, can be used to confirm a functional GT diagnosis. A full genomic sequence, including 45 exons and the associated splice sites, should be investigated to identify the responsible genetic lesion.1,3
1. Solh T, Botsford A, Solh M. Glanzmann's thrombasthenia: pathogenesis, diagnosis, and current and emerging treatment options. J Blood Med 2015;6:219-27.
2. Poon MC, Di Minno G, d'Oiron R, Zotz R. New insights into the treatment of Glanzmann thrombasthenia. Transfus Med Rev 2016;30:92-9.
3. Nurden AT, Pillois X, Wilcox DA. Glanzmann thrombasthenia: state of the art and future directions. Semin Thromb Hemost 2013;39:642-55.
4. Sebastiano C, Bromberg M, Breen K, Hurford MT. Glanzmann's thrombasthenia: report of a case and review of the literature. Int J Clin Exp Pathol 2010;3:443-7.