
Early diagnosis of FXIII congenital deficiency (FXIII CD) is essential to prevent potentially life-threatening bleeding episodes. Initial clot formation is not affected in FXIII CD, and intrinsic and extrinsic pathways function normally if there is no other concomitant coagulant disorder. Thus standard coagulation tests, including prothrombin time, activated partial thromboplastin time, fibrinogen, platelets and bleeding time are unaffected in individuals with FXIII CD.2-4 Therefore, a complete evaluation of the clotting system, including specific FXIII laboratory assays are required for the diagnosis of FXIII CD.
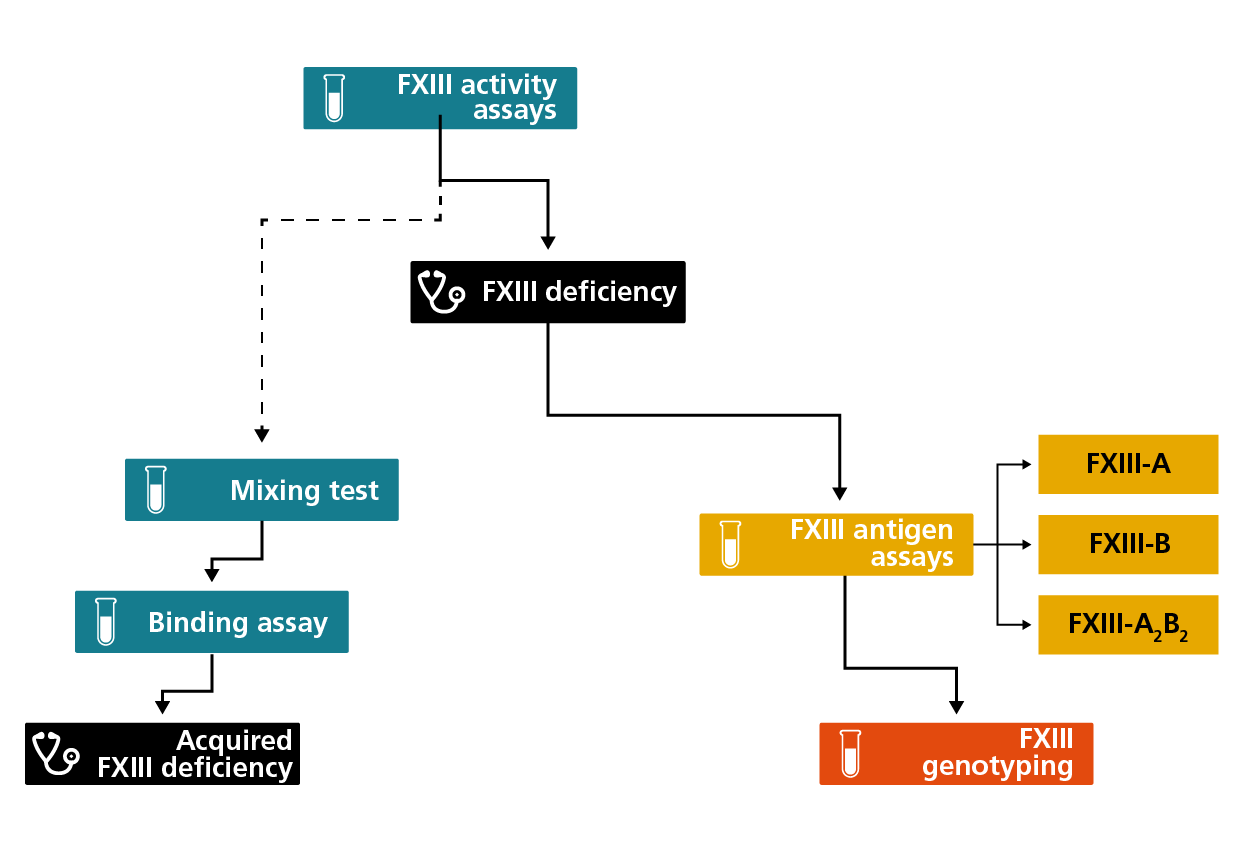
Algorithm for the diagnosis and classification of FXIII deficiencies, recommended by the Factor XIII and Fibrinogen SSC Subcommittee of the International Society on Thrombosis and Haemostasis (ISTH).2
Quantitative functional FXIII assays are the recommended first-line screening tests for patients with suspected FXIII CD.4 If a decrease in FXIII activity is detected, FXIII antigen assays should then be performed. Qualitative FXIII assays (i.e., clot solubility assays) are no longer recommended due to their low sensitivity and poor standardisation, however often these assays are the only available option in countries with restricted healthcare economies.4
Clot solubility assay
This qualitative method to screen for FXIII deficiency, which measures the solubility of fibrin clots in urea, acetic acid or monochloroacetic acid solution, was traditionally used and is still used in some regions with limited financial resources or where other FXIII assays are not available.1,2,4 However, this method is not recommended for FXIII deficiency screening because the assay only detects very severe FXIII deficiency with the limit of detection in the range of <0.5-5 IU/dL (%) FXIII activity.1,2,4,5 The assay is poorly standardised and its sensitivity depends on test conditions such as fibrinogen level, clotting reagent, solubilising agent and the concentration and time of the detection of solubility.1,2,4,5 The continued use of this test has contributed to the high number of missed or delayed FXIII deficiency diagnoses.1,2,4,5
Functional FXIII activity assays
FXIII activity assays are based on two principles: the measurement of ammonia release during the first step of the transglutaminase reaction and the measurement of labelled amine covalently cross-linked to a protein substrate by activated FXIII.1,2,4,5
The newer photometric assay is based on the isopeptidase activity of activated FXIII under certain conditions in which it can release primary amines bound to a glutamine residue in an oligopeptide.5
Once a diagnosis has been achieved, these quantitative functional assays can be used to monitor treatment with replacement factor concentrate.
Immunological FXIII antigen assays
Immunological FXIII antigen assays are based on the sandwich enzyme-linked immunosorbent assay (ELISA) principle. The advantage is their high sensitivity; the disadvantage is that they are more time-consuming and not easy to automate.4
Mixing tests
Mixing assays detect the presence of neutralising antibodies against FXIII-A which inhibit the FXIII activity of normal plasma when patient and normal plasma are mixed.2
Binding assays
Binding assays detect non-neutralising antibodies by measuring the binding of patients’ immunoglobulins to purified plasma FXIII and purified FXIII subunits in an ELISA or dot blot arrangement.2
Molecular analysis (genotyping)
Characterisation of the molecular genetic defect is performed in a research setting only and can identify causative genetic variants by complete sequencing of exonic and regulatory regions of the F13A1 and F13B genes.2,3
1. Bolton-Maggs PH, Favaloro EJ, Hillarp A, Jennings I, Kohler HP. Difficulties and pitfalls in the laboratory diagnosis of bleeding disorders. Haemophilia 2012;18 Suppl 4:66-72.
2. Kohler HP, Ichinose A, Seitz R, et al. Diagnosis and classification of factor XIII deficiencies. J Thromb Haemost 2011;9:1404-6.
3. Biswas A, Ivaskevicius V, Thomas A, Oldenburg J. Coagulation factor XIII deficiency. Diagnosis, prevalence and management of inherited and acquired forms. Hamostaseologie 2014;34:160-6.
4. Schroeder V, Kohler HP. Factor XIII deficiency: an update. Semin Thromb Hemost 2013;39:632-41.
5. Katona E, Penzes K, Molnar E, Muszbek L. Measurement of factor XIII activity in plasma. Clin Chem Lab Med 2012;50:1191-202.